Single cell transcriptomic analysis of the immune cell compartment in the human small intestine and in Celiac disease
Abstract
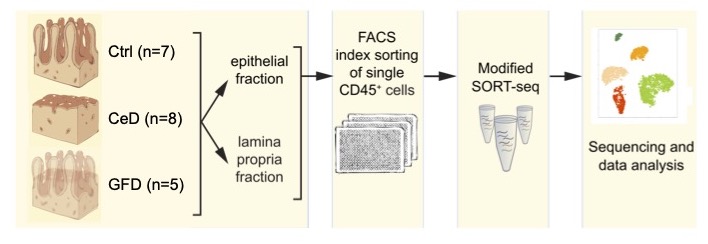
Celiac disease is an autoimmune disorder in which ingestion of dietary gluten triggers an immune reaction in the small intestine leading to destruction of the lining epithelium. Current treatment focusses on lifelong adherence to a gluten-free diet. Gluten-specific CD4+ T cells and cytotoxic intraepithelial CD8+ T cells have been proposed to be central in disease pathogenesis. Here we use unbiased single-cell RNA-sequencing and explore the heterogeneity of CD45+ immune cells in the human small intestine. We show altered myeloid cell transcriptomes present in active celiac lesions. CD4+ and CD8+ T cells transcriptomes show extensive changes and we define a natural intraepithelial lymphocyte population that is reduced in celiac disease. We show that the immune landscape in Celiac patients on a gluten-free diet is only partially restored compared to control samples. Altogether, we provide a single cell transcriptomic resource that can inform the immune landscape of the small intestine during Celiac disease.