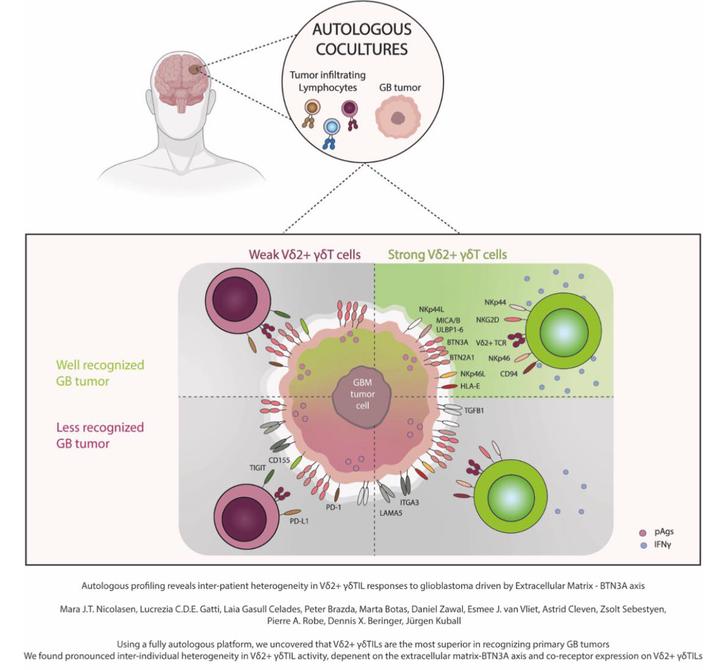
Autologous profiling reveals inter-patient heterogeneity in Vδ2+γδTIL responses to glioblastoma driven by extracellular matrix-BTN3A axis
Abstract
Background The effectiveness of immunotherapies against glioblastoma (GB) remains limited. A major obstacle in advancing new strategies is the reliance on non-autologous systems, which do not accurately mimic the true extent of inter-patient heterogeneity in both immune responses and tumor susceptibility. This often leads to misleading conclusions about therapeutic efficacy and targetability. Methods: In this study, we addressed this critical gap by employing a fully autologous model. We phenotypically characterized primary αβ and γδT cells from the peripheral blood and tumors of 40 brain tumor patients, including 36 with confirmed GB, and expanded and functionally assessed the autologous anti-GB reactivity in a subset of patients. Results: Notably, only Vδ2+ and Vδ2− γδT cells, but not αβT cells, recognized autologous tumors. While Vδ2− γδT cells showed activity in a subset of patients, Vδ2+ γδTILs from all patients responded to autologous GB cells in the presence of pamidronate. In patients, a higher percentage of Vδ2+ γδTILs was associated with longer overall survival. However, the potency of Vδ2+ γδTILs varied markedly between individuals, highlighting substantial inter-patient heterogeneity in γδT cell-mediated tumor recognition. This variability was driven by differences in both immune cell-intrinsic features and tumor-intrinsic factors, including expression of BTN2A1 and especially BTN3A, the ligands of the Vδ2+ γδTCR. Functional assays revealed that anti-GB reactivity was further modulated by stimulatory and inhibitory co-receptors such as NKG2D, CD94, and TIGIT. Transcriptomic analysis linked Vδ2+ γδT cell reactivity to extracellular matrix (ECM) pathways and disrupting ECM components such as LAMA5 and TGFB1 enhanced T cell responses. Knockout of ITGA3, a LAMA5 receptor, increased BTN2A1 and BTN3A expression on GB cells, improving immune recognition. Conclusions: This study demonstrates that inter-patient heterogeneity in Vδ2+ γδTIL responses to GB is driven by the extracellular matrix-BTN3A axis. Autologous systems effectively capture this heterogeneity, offering a reliable platform to identify determinants of both immune function and tumor vulnerability, insights that are essential for the rational design of γδTIL-based immunotherapies.