Live-cell fluorescence correlation spectroscopy dissects the role of coregulator exchange and chromatin binding in retinoic acid receptor mobility
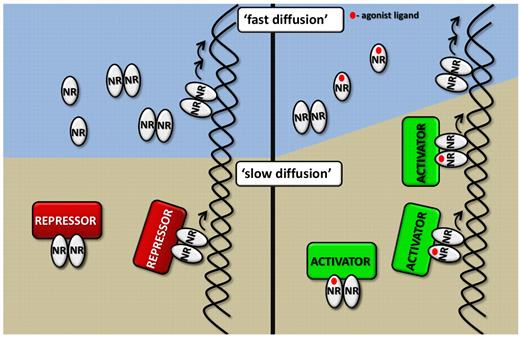 Interpretations of the different states and populations of RAR as detected by FCS. (see publication)
Interpretations of the different states and populations of RAR as detected by FCS. (see publication)
Abstract
The retinoic acid receptor (RAR) is a member of the nuclear receptor superfamily. This ligand-inducible transcription factor binds to DNA as a heterodimer with the retinoid X receptor (RXR) in the nucleus. The nucleus is a dynamic compartment and live-cell imaging techniques make it possible to investigate transcription factor action in real-time. We studied the diffusion of EGFP–RAR by fluorescence correlation spectroscopy (FCS) to uncover the molecular interactions determining receptor mobility. In the absence of ligand, we identified two distinct species with different mobilities. The fast component has a diffusion coefficient of D1=1.8–6.0 μm2/second corresponding to small oligomeric forms, whereas the slow component with D2=0.05–0.10 μm2/second corresponds to interactions of RAR with the chromatin or other large structures. The RAR ligand-binding-domain fragment also has a slow component, probably as a result of indirect DNA-binding through RXR, with lower affinity than the intact RAR–RXR complex. Importantly, RAR-agonist treatment shifts the equilibrium towards the slow population of the wild-type receptor, but without significantly changing the mobility of either the fast or the slow population. By using a series of mutant forms of the receptor with altered DNA- or coregulator-binding capacity we found that the slow component is probably related to chromatin binding, and that coregulator exchange, specifically the binding of the coactivator complex, is the main determinant contributing to the redistribution of RAR during ligand activation.